Questions 35-40 are based on the following passage.
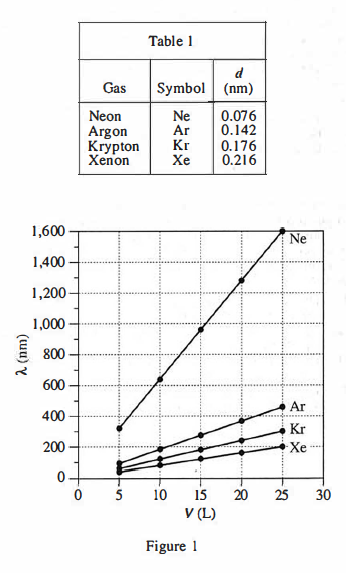
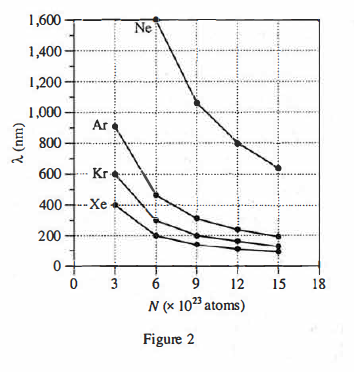
For gas atoms in a state of random motion, the mean free path, λ, is the average distance a gas atom will travel between collisions with other gas atoms. This distance depends upon the diameter of the gas atom, d, the volume of the gas, V, and the number of atoms of the gas, N. Table 1 lists the name, symbol, and value of d (in nanometers, nm) for each of 4 gases. Figure 1 shows, for each gas, at 293 kelvins (K), how A (in nm) varies with V (in liters, L) in a sample with N = 6 x 10²³ atoms of the gas. Figure 2 shows, for each gas, at 293 K, how A varies with N in a sample with V = 25 L.